 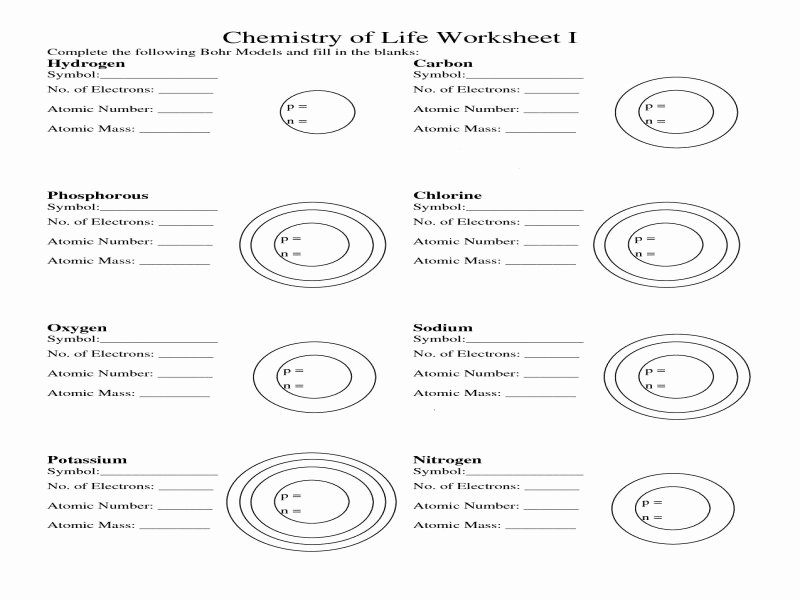
The Bohr model depicts an atom as a central nucleus containing protons and neutrons, with electrons in circular orbits at a certain distance from the crux. In 1913 the initial diagram of the atom was developed by a Danish scientist, Niels Bohr. When the electrical is neutral, each element has as many electrons as its atomic number. In all electrically neutral atoms, the number of electrons is the same as the number of protons. There is a correlation between the number of protons in an element, the atomic number that distinguishes one component from another, and the number of electrons it has. Niel Bohr presented this idea as a center of a nucleus that contains protons and neutrons while an electron is in shells orbiting them. The Bohr diagram, also known as the Bohr model, is an ancient model of a hydrogen atom. The Bohr model raises difficult concerns for scientists and philosophers alike, and it remains a watershed moment in scientific thought. The Bohr diagram evolved from a comparable model developed by J. Bohr Model of the Atom Worksheet Answers.Diagram of the First 20 Elements Bohr Model.Bohr diagram worksheets, whether as an object or subject, are a wonderful resource for both instructors and students. This engaging and participatory method not only improves their knowledge and comprehension but also helps to reinforce basic atomic structure ideas. Learners can practice drawing accurate Bohr diagrams, which show the arrangement of electrons in an atom's energy levels, by utilizing these worksheets. For pupils studying chemistry or physics, Bohr diagram worksheets can help them visualize and understand the structure of atoms.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
